COVID-19 vaccine now available for children 5 and older
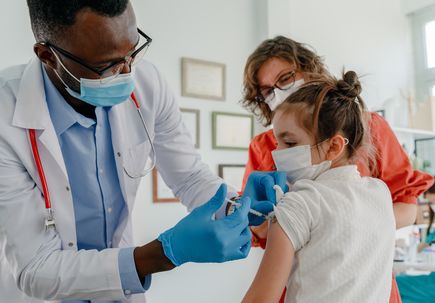
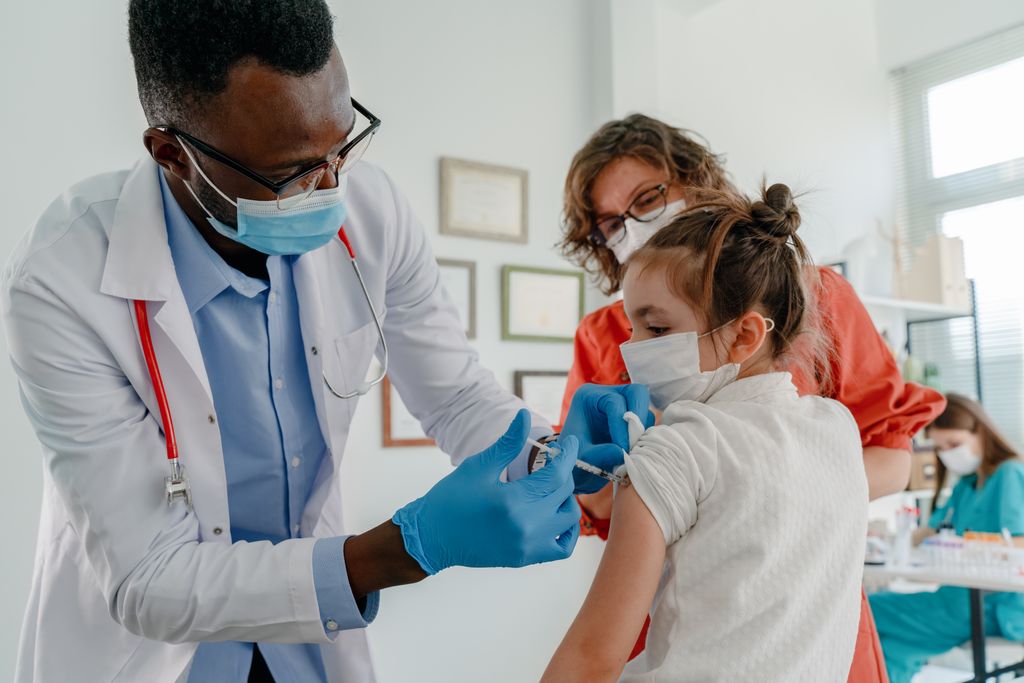
The Food and Drug Administration, Centers for Disease Control and Prevention and the Western States Scientific Safety Review Workgroup have authorized and recommended the Pfizer-BioNTech COVID-19 vaccine for children 5 to 11 years old. The vaccine previously received emergency use authorization for individuals 12 and older in May 2021.
“As both a parent and a pediatrician, I am grateful that we now have another tool to help protect our kids from the devastating impacts of COVID-19,” says Eliza Hayes Bakken, M.D., associate professor of pediatrics, OHSU Doernbecher Children’s Hospital.
“OHSU supports federal and state guidance to offer the Pfizer-BioNTech vaccine to eligible children ages 5 to 11, and believes that being vaccinated against COVID-19 is the single best thing people can do to help end the pandemic and keep our families, schools and communities safe. Based on available evidence, we know that the recommended dose of 10 micrograms is safe and effective in limiting severe illness, and that the benefits of the vaccine far outweigh the risks,” says Bakken.
What to know about COVID-19 vaccines and kids
- The Pfizer-BioNTech vaccine for children ages 5 to 11 is safe and effective: The vaccine was studied in approximately 3,100 children, and no serious side effects have yet been detected in the ongoing study. Additionally, the study found that the vaccine is more than 90% effective in preventing COVID-19 in kids.
- Doses of the Pfizer-BioNTech COVID-19 vaccine are smaller for kids than adults: The Pfizer-BioNTech vaccine for children 5 to 11 years old is the same vaccine available to people 12 and older. However, those younger than 12 will receive two doses of 10 micrograms, 21 days apart. Children 12 years old and older receive two doses of 30 micrograms, 21 days apart. The smaller doses are based on the development of the pediatric immune system, not a child’s size or weight.
- Vaccine side-effects are possible, but the benefits outweigh the risks: Side effects, such as headache, fatigue or pain at the injection site are common following most vaccines, including COVID-19. Although they may be uncomfortable, mild side effects show that the vaccine is working to build protection against a virus.
While there is no evidence to confirm long-term side effects from available COVID-19 vaccines, rare side effects from any vaccine or medication are always possible. A small number of heart inflammation, or myocarditis, cases have been reported in teenagers following COVID-19 vaccination. These cases are exceedingly rare, and experts suggest the risks of having myocarditis following COVID-19 infection is far greater than following vaccination.
- COVID-19 vaccines do not cause infertility or alter DNA: There is no evidence to suggest that available COVID-19 vaccines impact infertility or puberty. The vaccines also cannot cause COVID-19 disease or alter DNA. Experts will continue to monitor the COVID‑19 vaccines to ensure they are safe and effective.
- Although risk of serious illness from COVID-19 is low, kids should still get vaccinated: While data tell us that children are less likely than adults to contract serious illness from COVID-19, with the highly contagious delta variant, more children become sick from the virus and may be at risk for post-infection complications such as MIS-C. Information about the long-term impacts of the disease are still unclear.
Trial data show that the vaccine effectively decreases the risk of contracting COVID. As more members of our communities are vaccinated the transmission of this deadly virus will slow.
- People of all ages should consider vaccination even if they’ve previously had the virus: Research suggests that vaccines can provide a stronger COVID‑19 immune response than the one naturally present in people who had COVID‑19. It is possible that people can contract COVID-19 more than once; therefore, vaccines will help to limit risks of infection and spread.
- Everyone should continue to wear masks and physically distance even if they’ve been vaccinated: Vaccines are one of the best ways to help protect yourself and others from getting COVID-19; however, they are just one tool in our toolbox. It is important to take a layered approach to limiting virus spread, including vaccination, masking, hand washing and physical distancing.
In Oregon, masks are required for individuals 5 and older in indoor and many outdoor public settings, and for those 2 and older on public transit. OHSU recommends wearing a mask outdoors any time you can't stay 6 feet from others. The CDC has also recommended that teachers, staff, students and visitors to schools wear a mask indoors, regardless of vaccination status.
Learn more about kids and preparing for COVID-19 vaccines here.
People with questions about COVID-19 should contact their provider.
Vaccination information for individuals 12 years old and older, including third dose and booster shots, is available at www.ohsu.edu/covidvaccines.